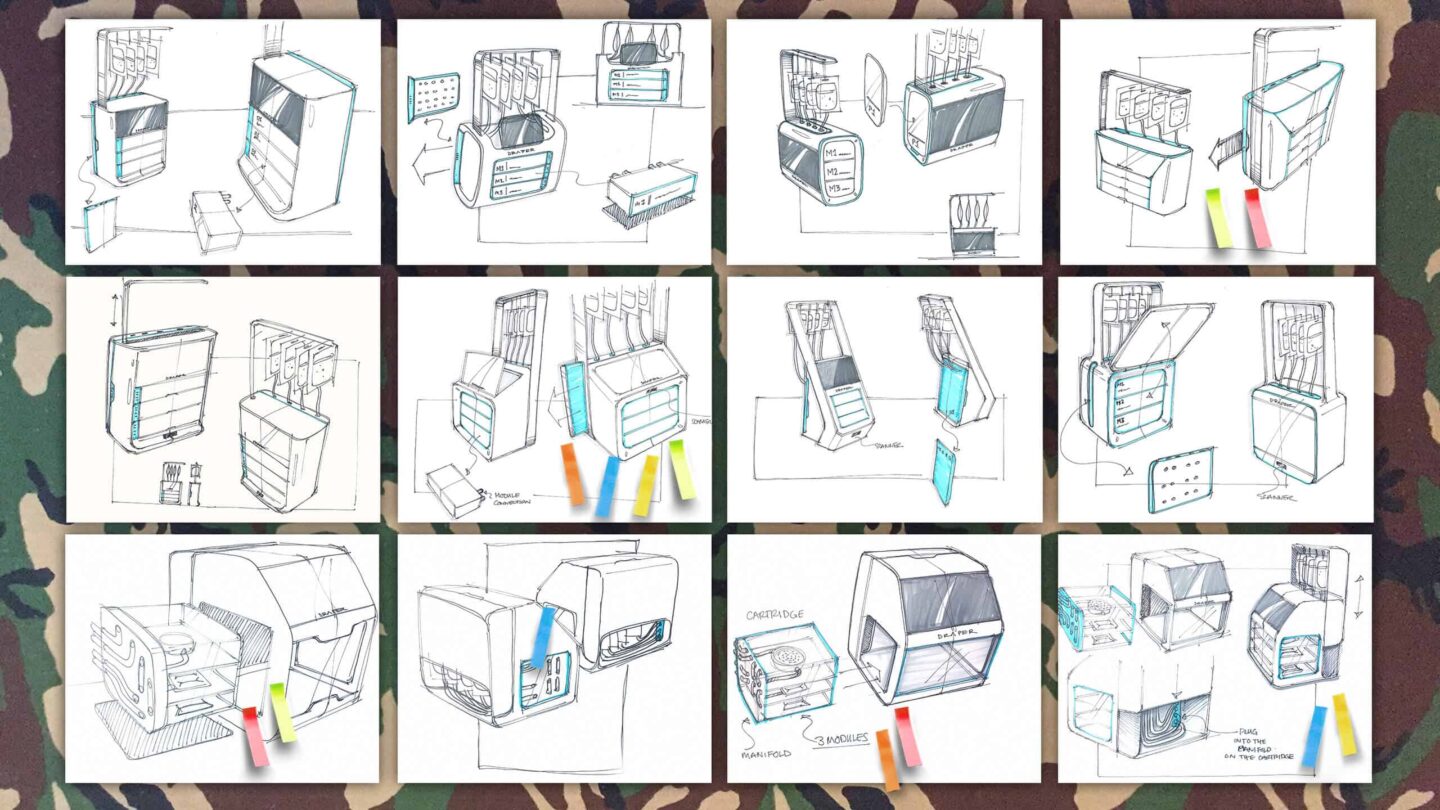
Immunotherapy Bioprocessing Device

Cell-based immunotherapies offer life-saving potential, but traditional bioprocessing is slow, inconsistent, and expensive. Draper aimed to design a modular system that could make production faster, more reliable, and accessible to more patients.
Read more
Sprout worked with Draper to translate complex microfluidic technology into a device that was functional, intuitive, and adaptable for clinical use. Our goal was to create a design that balanced advanced science with ease of operation, while supporting Draper’s broader vision for a unified product ecosystem.
Collaborating closely with Draper’s engineers, we developed the device’s internal and external design. A patient-specific cartridge system and customizable rear manifold allow flexibility across therapies. We refined the user experience with clear cues such as press points, lighting modes, and a streamlined interface, making the system intuitive to navigate and straightforward to operate. The system integrates alerts and real-time monitoring via push notifications and camera views, ensuring oversight at every step. Beyond this single device, the design establishes a visual brand language Draper can apply across different divisions and product categories.
The result is a bench-ready platform that reduces manual steps, improves reliability, and sets the stage for broader immunotherapy access. By pairing Draper’s pioneering science with Sprout’s design expertise, the device helps labs work more efficiently and supports Draper’s mission to bring breakthrough treatments to more patients.








View case study
How we designed three award-winning products that can save lives and change the world